The word collagen refers to a diverse family of glycoproteins found in the extracellular matrix (ECM). In all its forms, collagen is the most abundant protein in animals, making up approximately 25–30% of total protein mass (in humans, it corresponds to approximately 6% of total body weight). In vertebrates, it is the main constituent of skin, tendons, cartilage, and bone. [Exposito]
Collagen is insoluble in water and is also resistant to the in vitro action of digestive enzymes. Heat treatment and/or the action of acids, alkalis, or specific enzymes causes a partial degradation of collagen with cleavage of some covalent bonds, leading to the formation of gelatin, a heterogeneous mixture of peptides which is more soluble of the starting material. Gelatin is often used in foods, pharmaceuticals, photographic films and cosmetics. A further, more extensive enzymatic degradation of gelatin results in gelatin (or collagen) hydrolysates. [Shigemura4]
The term collagen derives from the Greek κόλλα, meaning “glue,” and the suffix -γέν, denoting “producing.” In fact, as early 3000 years ago, Egyptians used gelatin solutions as a glue to repair wooden articles. [Silvestrini]
Gelatin has been eaten for a long time. Aspic, which is produced by boiling fish heads, was introduced as a high-class dish in a cookbook in Baghdad approximately during the 10th century. The modern use of gelatin for desserts dates from the 18th century, when mass production became possible. [Kubo]
The structure of collagen
Composed of 19 of the 20 proteinogenic amino acids (with the exception of tryptophan1) and significant amounts of some unusual amino acids, including hydroxyproline (Hyp) and hydroxylysine (Hyl), collagen has a very robust structure that provides elasticity, firmness, and strength to all human tissues. The most abundant amino acids in collagen are glycine (Gly) and proline (Pro).
To date, 29 types of collagens have been identified [Shenoy] (v. tab. 1).
On the basis of their conformations, the currently identified collagens can generally be classified into one of the following three groups: fibrous (or fibrillar) collagen, fibril-associated collagen, and basement membrane collagen. [Gelse]
Fibril-forming (or fibrillar) collagens (called this way because they are capable of organizing into fibrils) and (non-fibrillar) type IV collagen (characteristic of the basement membrane of the ECM) have been described in all animals from sponges to humans. [Exposito, Sutmuller]
Fibril-forming collagens, such as types I and III, play a major role in the structural mechanics of human tissues. Type I collagen is the most abundant in the human body; type I and type III, both fibrillar, make up over 90% of human collagen. [Wikipedia]
|
Type |
Notes |
|
I |
This is the most abundant collagen of the human body. It is present in scar tissue, the end product when tissue heals by repair. It is found in tendons, skin, artery walls, cornea, the endomysium surrounding muscle fibers, fibrocartilage, and the organic part of bones and teeth. |
|
II |
Hyaline cartilage, makes up 50% of all cartilage protein. Vitreous humour of the eye. |
|
III |
This is the collagen of granulation tissue and is produced quickly by young fibroblasts before the tougher type I collagen is synthesized. Reticular fiber. Also found in artery walls, skin, intestines and the uterus |
|
IV |
Basal lamina; eye lens. Also serves as part of the filtration system in capillaries and the glomeruli of nephron in the kidney. |
|
V |
Most interstitial tissue, associated with type I, associated with placenta |
|
VI |
Most interstitial tissue, assoc. with type I |
|
VII |
Forms anchoring fibrils in dermoepidermal junctions |
|
VIII |
Some endothelial cells |
|
IX |
FACIT collagen, cartilage, assoc. with type II and XI fibrils |
|
X |
Hypertrophic and mineralizing cartilage |
|
XI |
Cartilage |
|
XII |
FACIT collagen, interacts with type I containing fibrils, decorin and glycosaminoglycans |
|
XIII |
Transmembrane collagen, interacts with integrin a1b1, fibronectin and components of basement membranes like nidogen and perlecan. |
|
XIV |
FACIT collagen, also known as undulin |
|
XV |
– |
|
XVI |
FACIT collagen |
|
XVII |
Transmembrane collagen, also known as BP180, a 180 kDa protein |
|
XVIII |
Source of endostatin |
|
XIX |
FACIT collagen |
|
XX |
– |
|
XXI |
FACIT collagen |
|
XXII |
FACIT collagen |
|
XXIII |
MACIT collagen |
|
XXIV |
– |
|
XXV |
– |
|
XXVI |
– |
|
XXVII |
– |
|
XXVIII |
– |
|
XXIX |
Epidermal collagen |
Table 1: Known collagen types and related tissues (adapted from [Shoulders, Wikipedia])
Structurally all types of collagens are made up of protein units, each consisting of three polypeptide chains (α chains), sometimes identical, that intertwine to form a structure containing at least one triple-helical domain. [Exposito]
The parts of the α chain involved in the formation of the triple-helical structure are composed of sequences of Gly-X-Y triplets (where Gly is a glycine and X and Y are two generic amino acids) forming the so-called collagenous domain ([Exposito]). The X and Y positions are most often occupied by proline (Pro) and hydroxyproline (Hyp), respectively ([Taga]).
In fibrillar collagens, the α chains typically consist of a central collagenous portion composed of an uninterrupted sequence of Gly-X-Y triplets that coils around itself to form a left-handed helix, flanked by two non-collagenous domains (i.e., not composed of Gly-X-Y sequences). The chains then assemble into groups of three, coiling together to form structures consisting of a single right-handed triple-helical domain flanked by two uncoiled portions (telopeptides). These structures form tropocollagen2. [Exposito, Quan]
Collagen biosynthesis
Collagen is synthesized by different cell types depending on the tissue (for example, fibroblasts in connective tissue, osteoblasts in bone). In particular, collagen biosynthesis occurs in several stages, some of which are intracellular and others extracellular.
For fibrillar collagen types, the process is schematically the following.
- The α chains (denoted as α1, α2, or α3) are formed first. They are synthesized by ribosomes located on the outer surface of the Rough Endoplasmic Reticulum (RER) as three-dimensional stranded pre-pro-peptides (prochains or preprocollagen). The pre-pro-peptides are subsequently released into the lumen of the RER where they are transformed in propeptides by cleavage of the N-terminal signal sequence (the first part of the protein strand). [Carlson]
- Within the RER, the propeptides are subjected, during their elongation, to some post-translational modifications: some lysines and prolines of the propeptides are hydroxylated by the enzymes lysyl hydroxylase and prolyl hydroxylase respectively, producing hydroxylysine (Hyl) and hydroxyproline (Hyp3). Both these enzymes require vitamin C as a cofactor4. This step helps in cross-linking the α chains and stabilize the triple helix, and it is the reason why collagen contains significant amounts of the unusual Hyp and Hyl aminoacids. While prolyl hydroxylation in the Y position is the norm, hydroxylation of prolines in the X-position is much less common, there being only one such residue per chain in collagens I and II, in contrast to collagen IV which has several. [Carlson, Harwood, Hulmes, Michelacci]
- The extent of post-translation modification of prolyl and lysyl residues in the diverse collagen types differs substantially. For example, the relative extent of lysyl hydroxylation in type IV collagen is very high (more than 90% of the residues at position Y are hydroxylated), while in types I and III only 15% are hydroxylated. [Michelacci]
- Hydroxylated lysine residues (but not Hyp) can be subsequently further modified by specific enzymes (galactosyl transferase and galactosylhydroxylysyl-glucosyl transferase5) of the endoplasmic reticulum which add galactose and glucose moieties to the hydroxy group of hydroxylysine residues, to form galactosylhydroxylysine and then glucosylgalactosylhydroxylysine (thus, both O-glycosides). [Canty, Carlson, Harwood]
- Furthermore, some more oligosaccharides may be present in the propeptide extensions. These oligosaccharides are synthesized on a dolichol lipid intermediate in the RER membrane and transferred intact to the pro-α chain. There is initial cleavage of the oligosaccharide while the chain is still in the RER, and further processing into the Golgi apparatus. [Michelacci]
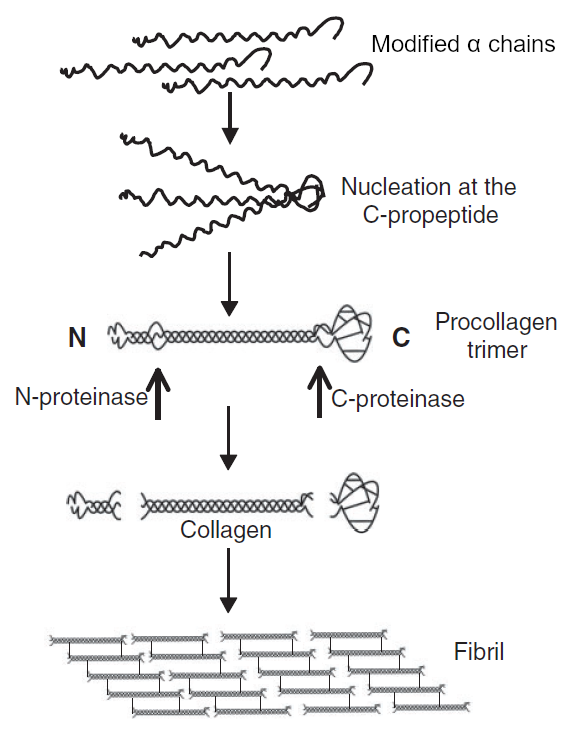
Fig. 1: Biosintesi del collagene, step 6-11 (adattato da [Canty])
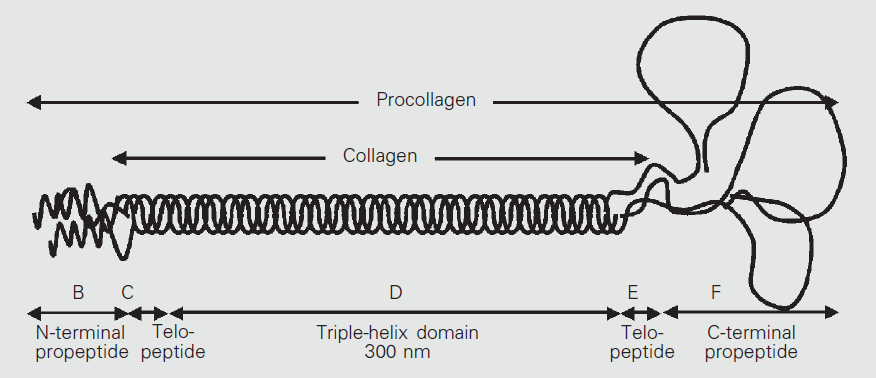
Fig. 2: Procollagene di tipo I (adattato da [Michelacci]) - After that, three of the modified α chains twist together forming procollagen (fig. 1,2). The newly-formed procollagen structure consists of a central right-handed helix (triple helical domain), about 300 nm in length, exclusively made of the (Gly-X-Y)n α chain sequences, flanked by two terminal globular, non-spiralized sequences which do not exhibit the Gly-X-Y repeat structure, one at each end of the chain (N- and C-propeptides). [Canty, Carlson, Hulmes]
- The formation of disulfide bonds between the C-terminal peptides of the α chains brings the three peptides into an arrangement favorable to the helix formation (nucleation). The folding of the collagen triple helix is directional and occurs in the C-to-N direction (fig. 1). The three α chains may either be identical (homotrimeric collagen, as in collagen III composed of three α1(III) chains) or different (heterotrimeric or heterotypic collagen, as in collagen I composed of two α1(I) and one α2(I) chains)6. [Canty, Carlson]
- Procollagen molecules are translocated to the Golgi apparatus where the oligosaccharides added in step 5 are trimmed and synthesis of a high-mannose structure occurs. In addition, phosphorylation of certain serine residues and sulfation of some tyrosine residues occur in some collagens. After that, procollagen is packaged in secretory vesicles that fuse with the cell membrane and release their contents into the extracellular environment (exocytosis). [Carlson, Michelacci]
- Outside the cell, membrane-bound enzymes (procollagen N- and C-proteinases) remove most part of the globular ends of the procollagen, leaving only two small residues (telopeptides) of the unwound portion at either end, producing tropocollagen, a molecule which therefore consists of a single triple helix domain flanked by the two short terminal telopeptide portions: N-telopeptide (located at the N-terminal end) and C-telopeptide (located at the C-terminal end of procollagen). The telopeptides comprise roughly 2-5% of the tropocollagen molecule, and have been shown to influence fibril assembly and stability (fig. 1,2). [Canty, Carlson]
- Once cleaved, tropocollagen molecules rapidly aggregate into ordered structures, arranging one after the other, orienting themselves thanks to the presence of telopeptides (the N-telopeptide opposes the C-telopeptide), and disposing themselves in parallel rows, forming string-like structures called fibrils (fig. 1).
- Within fibrils, the tropocollagen packing is characteristically staggered by a regular 1/4 molecular length, relative to adjacent molecules (fig. 1). Quarter-stagger packing and the whole process of fibril formation are nonenzymatic processes and are driven and stabilized by large numbers of noncovalent bonds on all sides of the packed rod molecules within each fibril, even if the nature of the interactions that govern fibril diameter is not completely understood. [Carlson, Michelacci]
- Collagen molecules in fibrillar array become substrate for the copper-dependent enzyme lysyl oxidase which oxidatively deaminates certain lysyl and hydroxylysyl residues, transforming their side chain (-CH2-NH2) amino group into an aldehyde group (-CHO), and thus converting lysine to allysine and hydroxylysine to hydroxyallysine. These reactive aldehyde groups condense with lysyl or hydroxylysyl residues in adjacent molecules to form covalent intermediate crosslinks, which can evolve, during collagen maturation, to histidine adducts or, by addition to a third aminoacidic residue, to pyrrolic or pyridinium compounds. These bonds are responsible for the tensile strength of collagen fibers. [Carlson, Exposito, Michelacci, Robins]
In collagen types IV, VI, VII, and XII (non-fibrillar), the triple-helical domains are interrupted by non-helical segments. [Exposito, Sutmuller]
Collagen digestion
Native collagen is almost resistant to isolated proteolytic enzymes, partly due to its insolubility, hence it is regarded as indigestible. However, denatured collagen can be acted upon by a number of proteolytic enzymes. Cooking and highly acidic conditions in the stomach (pH < 2.0) cause denaturation and unfolding of the triple helix of collagen, making it susceptible to enzymatic digestion. So, gelatin and gelatin hydrolysates prepared by partial hydrolysis of gelatin can be easily digested and absorbed by the small intestine. [Asghar, Cerda, Taga]
Nutritional trials with rats have indicated that the apparent digestibility7 of limed collagen8 from cattlehide was 90% [Asghar, Whitmore], and trials on human volunteers have shown that, although not all of the protein is absorbed, collagen9 is digested to the same extent as other proteins [Reuterswärd].
After the ingestion, proteins are broken down to smaller molecules before being absorbed from the gastrointestinal tract. In addition to individual free amino acids, some peptides and even macromolecules are absorbed intact from the gastrointestinal tract under normal conditions and transported into the bloodstream [Ichikawa, Iwai, Oesser, Ohara, Prockop, Shigemura, Shigemura3, Taga].
Several studies have been carried on animal intestinal tissues or intestine transport models (usually based on Caco-2 cells10) in order to understand how the different peptides are absorbed [Aito-Inoue, Matsui]. It is well-recognized that di- and tri-peptides can be transported through the brush border membrane peptide transporter PepT1, while the intestinal transport routes of oligopeptides with 4 or more aminoacidic residues remain unknown. As reported so far, the latter oligopeptides are mostly involved in passive diffusion routes via paracellular tight junctions. [Matsui, Xu]
In some ex vivo experiments on mice small intestine, peptides within a molecular weigth (MW) range from 1 kD to ~10 kD were found on the serosal side of the intestine after the application of a gelatin hydrolysate, indicating that a significant amount of the lysate can be absorbed in high-MW forms. [Oesser]
The peptides and macromolecular proteins absorbed intact from the gut retain some biological function, suggesting the possibility of a therapeutic effect for orally applied peptides and proteins [Oesser].
Collagen (and thus gelatin) is the only animal protein which contains significant amounts of hydroxyproline11. Essentially almost all the hydroxyproline in animal tissues and fluids is present as either a component of collagen or in the form of peptides or free hydroxyproline derived from the degradation of collagen. [Cerda]
After ingestion of gelatin by normal adult subjects, up to 8% of the hydroxyproline found in gelatin is absorbed in a peptide-bound form and then excreted in a peptide-bound form in urine. [Cerda, Prockop]
Over 50 years ago, Prockop et al. first reported that Hyp-containing peptides appear in blood at relatively high concentrations after oral ingestion of gelatin hydrolysate, although the peptide structure was not determined. [Prockop, Taga]
More recently, some researchers identified a group of Hyp-containing di- and tri-peptides in the blood of volunteers after the oral ingestion of gelatin hydrolysate and revealed that prolyl-hydroxyproline (Pro-Hyp) always accounts for a major portion of the collagen-derived Hyp-containing peptides, regardless of the animal source of the gelatin. Other Hyp-containing peptides were identified in the blood, including Ala-Hyp, Ile-Hyp, Leu-Hyp, Phe-Hyp, Hyp-Gly, Ala-Hyp-Gly, Pro-Hyp-Gly, and Ser-Hyp-Gly. The collagen-derived oligopeptides were detected in blood at significantly higher concentrations after the ingestion, probably because of their higher resistance to blood proteases. [Iwai, Ohara, Shigemura, Taga]
Some studies highlight that not all collagens are equivalent. For instance, Salmo salar fish skin collagen and bovine Achilles tendon collagen contains no cysteine at all, while small quantities are present in bovine femur collagen. Hydroxyproline content is 30% higher in bovine Achilles tendon than in fish skin, and femur collagen contains even more (about twice as fish skin). [Gauza-Włodarczyk]
|
Sequence |
porcine skin |
fish scalec |
fish skinc |
chicken feet |
chicken cartilage |
|
Ala-Hyp |
nda |
15% |
15% |
N/Ab |
N/A |
|
Ala-Hyp-Gly |
nd |
16% |
nd |
>3% |
>4% |
|
Ser-Hyp-Gly |
nd |
12% |
nd |
N/A |
N/A |
|
Pro-Hyp |
95% |
39% |
42% |
92% |
>70% |
|
Pro-Hyp-Gly |
nd |
5% |
3% |
>3% |
>19% |
|
Ile-Hyp |
>1% |
2% |
7% |
>1% |
>1% |
|
Leu-Hyp |
>3% |
10% |
27% |
>1% |
>5% |
|
Phe-Hyp |
>1% |
3% |
7% |
>1% |
>1% |
Table 2: Collagen peptide semi-quantitatively recovered in human plasma after oral ingestion of gelatin hydrolysates (anot detected, bdata not available, cfish species not known). Adapted from [Ichikawa, Iwai, Ohara].
Moreover, some Hyp-containing peptides appears in the blood only after the ingestion of gelatin produced with specific animals. In table 2 some examples are shown. [Ichikawa, Iwai, Ohara]
In a single study on rats, whole-body autoradiography at 30min after administration of oral [14C]-Pro-Hyp has shown a wide distribution of radioactivity in the body, including skin and articular cartilage, with the highest level of radioactivity observed in the gastric and intestinal walls. Incorporation of radioactivity into cells known to respond to Pro-Hyp such as dermal fibroblasts, synovial cells, chondrocytes, osteoblasts, and osteoclasts was observed. [Kawaguchi]
Analysis of the [14C]-Pro-Hyp metabolites in tissues and plasma showed that Pro-Hyp is partly distributed in the observed tissues in its intact form, which might be responsible for the biological functions of the dipeptide. [Kawaguchi]
In animals, most (nearly 90%) of the collagen derived trans-4-hydroxy-L-proline is catabolized to Gly via the trans-4-hydroxy-L-proline oxidase pathway, and trans-3-hydroxy-L-proline is degraded via the trans-3-hydroxy-L-proline dehydratase pathway to ornithine and glutamate, thereby conserving dietary and endogenously synthesized proline and arginine. [Hu]
Kinetics
Shigemura et al. verified the changes in the concentrations of free and peptide-incorporated Hyp over time in the plasma of human volunteers following the ingestion of a collagen hydrolysate prepared from cod (Gadus macrocephalus) skin gelatin by enzymatic hydrolysis. [Shigemura3]
The concentrations of both forms of Hyp began to increase at 0.5 h and reached a maximum level within 2 h after hydrolysate ingestion. The concentration of Hyp-containing peptides in plasma increased in a dose-dependent manner, reaching maximum levels of 6.43, 20.17 and 32.84 nmol/ml after ingestion of 30.8, 153.8, and 384.6 mg/kg doses, respectively, of collagen hydrolysate. Although the level of free Hyp decreased to two-thirds of its maximum 6 h after ingestion, the level was still significantly higher than before ingestion. At 4–6 h after ingestion, the level of Hyp-containing peptides returned to the pre-ingestion level. One-fourth of the maximum amount of total (free + peptide-incorporated) Hyp in plasma was in the form of Hyp-containing peptides. [Shigemura3]
The results showed that ingestion of more than 153.8 mg (per kg of body weight) of collagen hydrolysate significantly increased the levels of free and peptide-incorporated Hyp in plasma. However, ingestion of a 30.8 mg dose of collagen hydrolysate had no significant enhancing effect on the plasma level of either form of Hyp. [Shigemura3]
According to the authors, the results suggest that ingestion of a dose of only 30.8 mg of collagen hydrolysate might not be effective in promoting beneficial effects on skin, joints and blood lipid concentrations. No limit in the amount of Hyp that could be absorbed into the plasma was observed with ingestion of up to 384.6 mg of collagen hydrolysate per kg of body weight. So, ingestion of larger doses of collagen hydrolysate may enhance the distribution of food-derived Hyp-containing peptides in tissues and the consequent beneficial effects on the skin and joints. [Shigemura3]
Collagen and gelatin use in traditional and folk medicines
Gelatin is used in folk and traditional medicine in Asia since immemorial time. In Chinese Medicine, a special gelatin made with donkey hide (阿膠, E Jiao, Colla Corii Asini) is used to tonify and nourish the Blood, to stop bleeding, to nourish and moisten Yin, and to moisten the Lungs and Large Intestine. [AmDragon, Kubo, Shigemura4]
According to the Shennong Bencao Jing (Shennong’s Classic Materia Medica), compiled circa 1st to 2nd centuries ACE, E Jiao is also useful to prevent miscarriage and to stabilize the condition of unborn babies and pregnant women. [Kubo]
E Jiao was probably not made from donkey hide, as it is today, until the Tang dynasty (618–907 CE). Instead, the hide of other animals, primarily cows, was used. The primary raw material for E Jiao shifted from cowhide to donkey hide between the Tang Dynasty and the Song Dynasty (960–1276 CE). Today, oxhide gelatin is used to manufacture Huang Ming Jiao (黄明胶, Colla Corii Bovis), used to tonify Yin and moisten dryness, to stop bleeding, and to resolve swelling. [Kubo, TCMWiki]
Fish gelatin is used to manufacture Yu Biao (鱼鳔, or 鱼膠, Yu Jiao, Colla Pisces), used to nourish Yin, subdue Yang and nourish the Liver and Kidneys (in case of seminal emission, low libido, low back and knee weakness and soreness, cough), to stop bleeding, dissipate Blood Stasis and resolve swelling, and to calm the fetus. It is also used in case of tetanus and postpartum convulsions. [AmDragon]
Other kinds of gelatin are used in Chinese medicine, for example, tortoise shell gelatin (龟板胶, Gui Ban Jiao, Colla Plastrum Testudinis). Recently, pig skin gelatin (新阿胶, Xin E Jiao, “new” E Jiao) has been evaluated as a viable substitute for E Jiao [Zhao].
The medicinal use of animal gelatin is almost exclusive to China, Japan, and other East Asian countries. In Europe, Hildegard von Bingen, in her book “Physica” (dated around 1150 A.D.), recommended frequent and abundant meals of broth produced with veal fragments, very rich in collagen, to ease joint pain. [Kubo, Shigemura4, Silvestrini]
Modern uses of collagen and derivatives
The use of gelatin to facilitate the swallowing of medicine was invented by a French pharmacist, François Mothes, in the 19th century. In 1896, A. Dastre and N. Floresco gave dogs an intravenous injection of gelatin solution as a treatment to promote blood clotting without thrombosis. This is the earliest medicinal use of gelatin in the modern West. [Kubo]
Some in vivo studies on humans and animals have demonstrated that ingestion of gelatin has interesting health effects, for instance, leading to improvement of brittle nails and increase in hair diameter [Rosenberg, Scala, Shigemura4], enhancing non-heme iron absorption (by regulating systemic iron homeostasis rather than via solubilizing luminal ferric iron) [Wu], protecting from experimentally induced colitis [Zhu].
Recently, collagen peptides (aka, hydrolyzed collagen) have been proposed for the treatment of several health issues as well as for aesthetic purposes. Through hydrolysis, the long insoluble chains of collagen amino acids are broken down into smaller peptides that are soluble in water and assumed to be more digestible and better absorbed than both collagen and gelatin.
The first therapeutic application of hydrolyzed collagen dates back to the second half of the 20th century, when in the United States it was mainly used to treat affections of the skin appendages, which typically concerned nail weakness, improving nail growth and strength. [Silvestrini]
The ability of hydrolyzed collagen to improve the elasticity of tendons and ligaments, reduce joint pain and increase skin tone is now widely recognized (and advertised).
Some of the health effects of the ingestion of gelatin and hydrolyzed collagen are attributed to Gly (a conditionally essential amino acid with anti-inflammatory, immunomodulatory, and cytoprotective activities), Hyp and Hyp-containing oligopeptides, especially Pro-Hyp, the major degradation products of dietary gelatin entering the circulation [Cerda, Wu].
Hydrolyzed collagen: in vitro studies
Recent in vitro studies have shown that Hyp-containing peptides have several interesting physiological functions [Minaguchi, Nakatani, Ohara2, Shigemura, Shigemura2, Taga, Wu]. They have been found to be able to:
- stimulate the growth of skin fibroblasts and the production of collagen (mainly Pro-Hyp and Hyp-Gly) [Nakatani, Ohara2, Shigemura, Shigemura2, Shigemura3, Taga], a process known to be regulated by the presence of ascorbic acid [Murad, Taga];
- to promote the differentiation of osteoblasts, myoblasts, chondrocytes, preadipocytes and T-helper cells [Wu];
- to increase the synthesis of hyaluronic acid in fibroblasts, chondrocytes, and synovium cells [Wu];
- to inhibit the mineralization of chondrocytes [Nakatani, Shigemura3].
These facts suggest that the Hyp-containing collagen peptides might be associated to wound healing and inflammation process [Iwai].
Pro-Hyp-Gly, Pro-Hyp, and some other Hyp-containing peptides have chemotactic activity on fibroblast and peripheral blood neutrophils and monocytes [Ichikawa, Iwai, Laskin, Postlethwaite, Postlethwaite2]. Ala-Hyp and Gly-Pro-Val are potential inhibitors of angiotensin-converting enzyme [Kim, Iwai, Oshima].
Unfortunately, only a few studies compared the effects of Hyp-containing peptides with a mixture of the costituent amino acids as a control. In a couple of studies, Pro-Hyp effects have been compared with either Pro and Hyp alone and/or a mixture of Pro and Hyp and neither the amino acids alone nor their mixture were found able to produce the same effects of the Pro-Hyp dipeptide. [Minaguchi, Nakatani]
Anyhow, supplementation of Pro alone has been found unable to increase collagen production in human mammary CAFs (Carcinoma-Associated Fibroblasts) and human skin fibroblasts unless glutamine (Gln) availability or Pro synthesis12 is limited. Furthermore, although exogenous Pro increases COL1A113 expression and radiolabelled Pro is incorporated into collagen in human dermal fibroblasts, this effect is more pronounced when the fibroblasts are cultured in the absence of Gln. Therefore, it seems that fibroblasts preferentially synthesise their own Pro. This suggests that Pro availability determines the rate of collagen biosynthesis, while demand for Pro in fibroblasts is predominantly met by conversion from Gln. [Karna, Kay, Szoka]
In a single experiment, two weeks administration of oral Hyp in doses 0.5 or 1 g/kg doses to male rats increased the soluble collagen content of the skin, suggesting that oral Hyp administration augmented collagen production. [Aoki]
Hydrolyzed collagen: in vivo studies
The effects of the ingestion of hydrolyzed collagen by animals or human volunteers has been examined in some studies.
In a preclinical trial, the effects of the daily intake of 7 g of a commercially available collagen hydrolysate mixture containing 5 g of fish type I collagen hydrolysates on the skin properties of 25 Japanese female volunteers who tended to have dry and rough skin in winter were investigated. After 6 weeks of ingestion, the moisture contents in the stratum corneum of the face cheek, forearm, and back of the neck increased, suggesting skin condition improved. The viscoelastic properties including pliability and elasticity also significantly improved. Microscopic analysis also showed a significant improvement in the skin surface properties of the face cheek, such as greater smoothness, fewer wrinkles, and less roughness. Moreover, the skin surface pH remained unchanged at a weakly acidic pH of 5.9 during the whole study period, indicating the maintenance of good skin conditions. [Matsumoto]
In a placebo-controlled trial, the effects of daily ingestion of 2.5 or 5 g of porcine type I collagen hydrolysates on the skin parameters for cutaneous aging of 69 women aged 35 to 55 years were examined. After 8 weeks of ingestion, a significant increase in skin elasticity was observed in both dosage groups compared to the placebo-controlled group. At 4 weeks after the last ingestion for the treatment group, a longer lasting positive physiological effect on skin elasticity was still observed with the elderly women over 50 years old. Moreover, a positive effect of collagen hydrolysate ingestion on skin moisture and evaporation was also observed with the elderly women. [Proksch]
In an animal experiment with mice, the effect of daily ingestion of collagen peptide (0.2 g/kg body weight) prepared from fish scales on the skin damage induced by repeated UV-B irradiation was examined. Ingestion of collagen peptide suppressed UV-B-induced decreases in skin hydration, hyperplasia of the epidermis, and decreases in soluble type I collagen. [Tanaka]
In order to investigate the effects of collagen peptide ingestion on fibroblasts and the extracellular matrix in the dermis, collagen peptide was administered orally to pigs at 0.2 g/kg body weight/d for 62 d, and its effects were compared with those of lactalbumin and water controls. Fibroblast density, and diameter and density of collagen fibrils were significantly larger in the collagen peptide group than in the lactalbumin and water control groups. The two major components of dermal glycosaminoglycans, hyaluronic acid and dermatan sulfate, which are present in the inter-fibrillar space, did not differ significantly among the three groups. However, the ratio of dermatan sulfate, which is derived from fibril-bound decorin, was largest in the collagen peptide group. These results suggest that ingestion of collagen peptide induces increased fibroblast density and enhances formation of collagen fibrils in the dermis in a protein-specific manner. [Matsuda]
Hydrolyzed collagen for aesthetic purposes
For aesthetic purposes, hydrolyzed collagen is usually promoted as a supplement mainly aimed at counteracting the signs of aging.
With age, collagen production decreases while its degradation increases. This is caused by internal factors (hormonal and metabolic) or external factors, such as continued exposure to UV rays and pollution.
The decrease in collagen in our body is among the causes of:
- loss of skin tone;
- tendency to muscle mass reduction (sarcopenia);
- bone fragility (osteoporosis);
- cartilage pain and degeneration (arthritis). [Meritene]
The intake of hydrolyzed collagen is suggested as useful for attenuating all these processes typical of aging and, in particular, for:
- improve skin elasticity, tone and firmness;
- reduce joint and bone pain from arthritis and osteoarthritis;
- help maintain and develop muscle mass by supporting the fight against sarcopenia;
- strengthen nails and hair. [Meritene]
As we have seen (see paragraphs above), research seems to mostly confirm the claims above. Anyhow, it must be said that most of the studies done so far have been either carried out in vitro or in vivo on animals, or on small cohorts of human volunteers for relatively short periods of time. Moreover, the doses, the sources (bovine, porcine, marine, …) and the molecular weights of the peptides used in experiments vary markedly among the studies, making the conditions too heterogeneous for consistent results be drawn. Furthermore, last but not least, many studies are industry sponsored, especially the ones regarding the purported aesthetic effects.
Marine and mammalian collagen
The raw collagen used to produce hydrolyzed collagen is largely derived from slaughterhouse waste (hides, bones, ligaments, cartilage, etc.) from animals such as cattle, pigs, and horses. Many manufacturers, however, prefer to use the so-called marine collagen, obtained from fish processing waste.
Products made with plant-based collagen or vegan collagen are sometimes found on the market, but in reality, these products contain only substances or mixtures of substances that mimic collagen in consistency (especially in external cosmetic products) or composition, since collagen does not exist in the plant kingdom.
In animal skins and fish waste, the collagen type is predominantly type I; in bones, ligaments, and cartilage from mammals, varying amounts of type II and III collagen are also found.
Type I collagen is the most common type of product used for health care and skin beauty, while type II collagen is preferred and more used for joint health. [Campos]
Moreover, manufacturers often go so far as to explicitly tell the consumer that some specific collagen is better suited than another for a specific purpose, i.e., hydrolyzed type I collagen or hydrolyzed fish collagen “do more good” to the skin because skin is mostly composed of type I collagen (see for example box 1), but the reason behind this claim is reasonably quite weak: there is no reason for a given type of collagen derivative, once digested by the gastrointestinal enzymes and eventually by blood proteases (see, for example, [Matsui]), to be recycled by the body to reconstitute the same type of collagen (or any collagen at all).
|
Hydrolyzed marine collagen contains primarily Type I collagen. Hydrolyzed bovine collagen, on the other hand, contains Type II and Type III collagen. For this reason, marine collagen is commonly used to improve skin, slow aging, and support the mobility of tendons and ligaments. Hydrolyzed bovine collagen, on the other hand, may be recommended for cartilage problems, arthritis, or muscle strengthening. |
Box 1: A claim about the difference between fish and cattle hydrolyzed collagen [Meritene]
Several in vitro or ex vivo works have been carried out to study the gastrointestinal absorbability of peptides (see paragraph “Collagen digestion”), but the only molecules which are absorbed by the human body (and so can exert biological functions) for sure are the ones found in vivo in blood samples after (hydrolyzed) collagen ingestion (also see [Matsui]).
The free amino acids and peptides found in blood after consuming collagen derivatives vary significantly among animal sources and animal parts also for the same collagen type (for example, see tab. 2). The main peptides known to stimulate fibroblasts in vitro are Pro-Hyp and Hyp-Gly, major peptides found in blood after eating collagen from different sources14, meaning that collagen from any source can positively impact on skin health.
Indeed, the (mostly) type I marine collagen alone has been found able, either ex vivo, in vitro, or in vivo on human volunteers or on animals, to facilitate wound healing (see, for example, [de Souza]), to modulate cartilage metabolism [Boonmaleerat], and to positively impact on bone regeneration [Yamada], activities that in the human body also involve type II and type III collagen.
Claims about a better or faster absorbability of fish collagen (see for example box 2) have no real values, too, at least when we are dealing with hydrolyzed products (the absorption rate, here, depends on several factors, among which the mean MW of the peptides obtained by hydrolysis).
Marine collagen offers some real advantages over mammalian collagen.
First, marine collagen can be consumed even by people who, for cultural or religious reasons, cannot eat meat in general or certain types of meat (e.g., pork or beef). Second, marine collagen is not a potential source of transmission of diseases that may be of concern to human health (such as spongiform encephalopathies or avian influenza).
|
Fish collagen is absorbed up to 1.5 times more efficiently into the body, meaning that it has superior bioavailability over bovine or porcine types. Because it is absorbed more efficiently and enters the bloodstream more quickly, it is considered to be the best source of collagen for pharmaceutical applications. |
Box 2: A claim about the absorbability of fish collagen [Sripriya]
Furthermore, animal slaughter waste is often used as raw materials by the tanning and food industries (e.g., for the preparation of jellied meat or ready-to-use gelatins for food preparations), while fish processing waste is normally considered a by-product of little or no industrial importance. To date, fish waste is partly destined for the production of fishmeal, fertilizers, and fish oil with low profitability or utilized as raw material for direct feeding in aquaculture, but a huge amount of waste is thrown away [Coppola, Guillen]. This may impact on the price and availability of raw materials.
On the other hand, the majority of fish collagen contains less Pro and Hyp than mammalian-based collagen. [Oslan]
Additionally, fish collagen may have a residual fishy odor if not processed properly. [WangX]
Collagen toxicity
Hydrolyzed collagen is a safe material for both internal and external (i.e,. cosmetic) use, with a weak antigenicity. It was practically nontoxic when administered orally or dermally in acute animal toxicity studies. This ingredient was minimally irritating to rabbit eyes when tested full-strength. Primary skin irritation tests in rabbits indicated that hydrolyzed collagen was nonirritating or minimally irritating when tested full-strength. Subchronic dermal studies on two cosmetic formulations containing 2% hydrolyzed collagen were negative for systemic toxicity. Hydrolyzed collagen was nonsensitizing in guinea pigs. In clinical studies, hydrolyzed collagen produced no skin irritation, sensitization, or indication of phototoxicity. [Aguirre-Cruz, CIR]
References
|
[Aguirre-Cruz] |
Aguirre-Cruz, G. et al.,“Collagen Hydrolysates for Skin Protection: Oral Administration and Topical Formulation”, Antioxidants (Basel). 2020 Feb 22;9(2):181. doi: 10.3390/antiox9020181. |
|
[Aito-Inoue] |
Aito-Inoue, M. et al., “Transport of a tripeptide, Gly-Pro-Hyp, across the porcine intestinal brush-border membrane”, J Pept Sci. 2007 Jul;13(7):468-74. doi: 10.1002/psc.870. |
|
[AmDragon] |
American Dragon, https://www.americandragon.com/ |
|
[Aoki] |
Aoki, M. et al., “Increasing effect of an oral intake of L-hydroxyproline on the soluble collagen content of skin and collagen fragments in rat serum”; Biosci Biotechnol Biochem. 2012;76(6):1242-4. doi: 10.1271/bbb.120058. |
|
[Asghar] |
Asghar, A., Henrickson, R. L., “Chemical, biochemical, functional, and nutritional characteristics of collagen in food systems”, Adv Food Res. 1982:28:231-372. doi: 10.1016/s0065-2628(08)60113-5. |
|
[Boonmaleerat] |
Boonmaleerat, K. et al., “Fish Collagen Hydrolysates Modulate Cartilage Metabolism”, Cell Biochem Biophys. 2018 Jun;76(1-2):279-292. doi: 10.1007/s12013-017-0817-2. |
|
[Campos] |
Patrícia Maria Berardo Gonçalves Maia Campos et al.,“Oral Supplementation with Hydrolyzed Fish Cartilage Improves the Morphological and Structural Characteristics of the Skin: A Double-Blind, Placebo-Controlled Clinical Study”, Randomized Controlled Trial Molecules. 2021 Aug 12;26(16):4880. doi: 10.3390/molecules26164880. |
|
[Canty] |
Canty E. G. et al., “Procollagen trafficking, processing and fibrillogenesis”, J Cell Sci. 2005 Apr 1;118(Pt 7):1341-53. doi: 10.1242/jcs.01731. doi: 10.1242/jcs.01731. |
|
[Carlson] |
Carlson, B. M., “Chapter 2 – Tissues”. doi: 10.1016/B978-0-12-804254-0.00002-8. In: “The Human Body – Linking Structure and Function” (2018), ISBN: 978-0-12-804254-0, doi: 10.1016/C2015-0-00205-2. |
|
[Cerda] |
Cerda J. J. et al., “Intraduodenal hydrolysis of gelatin as a measure of protein digestion in normal subjects and in patients with malabsorption syndromes”, Gastroenterology. 1968 Mar;54(3):358-65. doi: 10.1016/S0016-5085(68)80096-4]. |
|
[CIR] |
CIR, “7 Final Report on the Safety Assessment of Hydrolyzed Collagen”, Cosmetic Ingredient Review. J. Am. Coll. Toxicol. 1985;4:199–221. doi: 10.3109/10915818509078691. |
|
[Coppola] |
Coppola, D. et al.,“Fish Waste: From Problem to Valuable Resource”, Mar Drugs. 2021 Feb 19;19(2):116. doi: 10.3390/md19020116. |
|
[de Souza] |
de Souza A. et al., “Fish collagen for skin wound healing: a systematic review in experimental animal studies”, Cell Tissue Res. 2022 Jun;388(3):489-502. doi: 10.1007/s00441-022-03625-w. |
|
[Exposito] |
Jean-Yves Exposito et al, “The Fibrillar Collagen Family”, Int. J. Mol. Sci. 2010, 11, 407-426. doi: 10.3390/ijms11020407. |
|
[Gauza-Włodarczyk] |
Gauza-Włodarczyk, M. et al., “Amino acid composition in determination of collagen origin and assessment of physical factors effects”, Int J Biol Macromol. 2017 Nov;104(Pt A):987-991. doi: 10.1016/j.ijbiomac.2017.07.013. |
|
[Gelse] |
Gelse, K. et al., “Collagens–structure, function, and biosynthesis”, Adv Drug Deliv Rev. 2003 Nov 28;55(12):1531-46. doi: 10.1016/j.addr.2003.08.002. |
|
[Guillen] |
Guillen, J. et al.,“A Review of the European Union Landing Obligation Focusing on Its Implications for Fisheries and the Environment”, Sustainability 2018, 10(4), 900. doi: 10.3390/su10040900. |
|
[Harwood] |
Harwood, R. et al., “Studies on the glycosylation of hydroxylysine residues during collagen biosynthesis and the subcellular localization of collagen galactosyltransferase and collagen glucosyltransferase in tendon and cartilage cells”, Biochem J. 1975 Nov;152(2):291-302. doi: 10.1042/bj1520291. doi: 10.1042/bj1520291. |
|
[Hu] |
Hu, S. et al., “Hydroxyproline in animal metabolism, nutrition, and cell signaling”, Amino Acids. 2022 Apr;54(4):513-528. doi: 10.1007/s00726-021-03056-x |
|
[Hulmes] |
Hulmes D.J.S. “Collagen diversity, synthesis and assembly”, In: “Collagen – Structure and Mechanics”. Mech. Fratzl P., editor. Springer Science+Business Media; 2008.; pp. 15–47. doi: 10.1007/978-0-387-73906-9_2. |
|
[Karna] |
Karna, E. et al., “Proline-dependent regulation of collagen metabolism”, Cell. Mol. Life Sci. 77, 1911–1918 (2020). doi: 10.1007/s00018-019-03363-3 |
|
[Kawaguchi] |
Kawaguchi T. et al, “Distribution of Prolylhydroxyproline and Its Metabolites after Oral Administration in Rats”, Biological and Pharmaceutical Bulletin, 2012, 35 (3), Pages 422-427. doi: 10.1248/bpb.35.422. |
|
[Kay] |
Kay EJ et al., “Regulation of Extracellular Matrix Production in Activated Fibroblasts: Roles of Amino Acid Metabolism in Collagen Synthesis”. Front. Oncol., 2021, 11:719922. doi: 10.3389/fonc.2021.719922 |
|
[Kim] |
Kim, S. K. et al.,“Angiotensin I converting enzyme inhibitory peptides purified from bovine skin gelatin hydrolysate”, J Agric Food Chem. 2001 Jun;49(6):2992-7. doi: 10.1021/jf001119u. |
|
[Kubo] |
Kubo, T. et al., “History of the Chinese Medicinal Gelatin”, Chinese Medicine and Culture (2022) 5:1. doi: 10.1097/MC9.0000000000000005. |
|
[Ichikawa] |
Ichikawa, S. et al., “Hydroxyproline-containing dipeptides and tripeptides quantified at high concentration in human blood after oral administration of gelatin hydrolysate”, Int J Food Sci Nutr. 2010 Feb;61(1):52-60. Doi: 10.3109/09637480903257711. |
|
[Iwai] |
Iwai, K. et al., “Identification of food-derived collagen peptides in human blood after oral ingestion of gelatin hydrolysates”. J. Agric. Food Chem. 2005, 53, 6531−6536. doi: 10.1021/jf050206p. |
|
[Laskin] |
Laskin, D. L. et al., “Chemotactic activity of collagen-like polypeptides for human peripheral blood neutrophils”, J Leukoc Biol. 1986 Mar;39(3):255-66. doi: 10.1002/jlb.39.3.255. |
|
[Matsuda] |
Matsuda, N. et al., “Effects of ingestion of collagen peptide on collagen fibrils and glycosaminoglycans in the dermis”, J Nutr Sci Vitaminol (Tokyo). 2006 Jun;52(3):211-5. doi: 10.3177/jnsv.52.211. |
|
[Matsui] |
Matsui, T., “Are Peptides Absorbable Compounds?”, J Agric Food Chem. 2018 Jan 17;66(2):393-394. doi: 10.1021/acs.jafc.7b05589. |
|
[Matsumoto] |
Matsumoto, H. et al., “Clinical effect of fish type I collagen hydrolysate on skin properties”. ITE Lett. 2006, 7:386–90 |
|
[Meritene] |
[https://www.meritene.it/salute/collagene-idrolizzato-a-cosa-serve] (Retrieved: 2025-04-15) |
|
[Michelacci] |
Michelacci, Y.M., “Collagens and proteoglycans of the corneal extracellular matrix”, Braz J Med Biol Res 36 (8), Aug 2003. doi: 10.1590/S0100-879X2003000800009. |
|
[Minaguchi] |
Minaguchi, J. Et al., “Effects of collagen-derived oligopeptide prolylhydroxyproline on differentiation of mouse 3T3-L1 preadipocytes”, Food Sci. Technol. Res. 2012, 18, 593−599. doi: 10.3136/fstr.18.593. |
|
[Murad] |
Murad, S. et al., “Regulation of collagen synthesis by ascorbic acid”, Proc. Natl. Acad. Sci. U.S.A. 1981, 78, 2879−2882. doi: 10.1073/pnas.78.5.2879. |
|
[Nakatani] |
Nakatani, S. et al., “Chondroprotective effect of the bioactive peptide prolyl-hydroxyproline in mouse articular cartilage in vitro and in vivo”, Osteoarthritis Cartilage 2009, 17, 1620−1627. doi: 10.1016/j.joca.2009.07.001. |
|
[Oesser] |
Oesser, S. et al., “Oral Administration of 14C Labeled Gelatin Hydrolysate Leads to an Accumulation of Radioactivity in Cartilage of Mice (C57/BL)”, J Nutr. 1999 Oct;129(10):1891-5. doi: 10.1093/jn/129.10.1891. |
|
[Ohara] |
Ohara H. et al., “Comparison of Quantity and Structures of Hydroxyproline-Containing Peptides in Human Blood after Oral Ingestion of Gelatin Hydrolysates from Different Sources”, J. Agric. Food Chem. 2007, 55, 1532-1535. doi: 10.1021/jf062834s. |
|
[Ohara2] |
Ohara, H. et al., “Collagen-derived dipeptide, prolinehydroxyproline, stimulates cell proliferation and hyaluronic acid synthesis in cultured human dermal fibroblasts”, J. Dermatol. 2010, 37, 330−338. doi: 10.1111/j.1346-8138.2010.00827.x. |
|
[Oshima] |
Oshima, G. et al, “Peptide inhibitors of angiotensin I-converting enzyme in digests of gelatin by bacterial collagenase”, Biochim Biophys Acta; 1979 Jan 12;566(1):128-37. doi: 10.1016/0005-2744(79)90255-9. doi: 10.1016/0005-2744(79)90255-9. |
|
[Oslan] |
Oslan, S. N. H. et al.,“Extraction and Characterization of Bioactive Fish By-Product Collagen as Promising for Potential Wound Healing Agent in Pharmaceutical Applications: Current Trend and Future Perspective”, Int J Food Sci. 2022 May 6:2022:9437878. doi: 10.1155/2022/9437878. |
|
[Postlethwaite] |
Postlethwaite, A. E. et al., “Chemotactic attraction of human fibroblasts to type I, II, and III collagens and collagen-derived peptides”, Proc. Natl. Acad. Sci. U.S.A. 1978,75, 871-875. doi: 10.1073/pnas.75.2.871. |
|
[Postlethwaite2] |
Postlethwaite, A. E. et al., “Collagen-and collagen peptide-induced chemotaxis of human blood monocytes”, J Exp Med. 1976 Jun 1;143(6):1299–1307. doi: 10.1084/jem.143.6.1299. |
|
[Prockop] |
Prockop, D. J. et al., “Gastrointestinal absorption and renal excretion of hydroxyproline peptides”. Lancet 1962, 2, 527−528: doi: 10.1016/S0140-6736(62)90400-2. |
|
[Proksch] |
Proksch E. et al.,“Oral supplementation of specific collagen peptides has beneficial effects on human skin physiology: a double-blind, placebo-controlled study”, Skin Pharmacol Physiol. 2014;27(1):47-55. doi: 10.1159/000351376. |
|
[Quan] |
Bryan D. Quan, Eli D. Sone, “Chapter Nine – Cryo-TEM Analysis of Collagen Fibrillar Structure”, Methods in Enzymology, Volume 532, 2013, Pages 189-205. doi: 10.1016/B978-0-12-416617-2.00009-6. |
|
[Reuterswärd] |
Laser Reuterswärd, A., Andersson, H., Asp N. G., “Digestibility of collagenous fermented sausage in man”, Meat Sci. 1985;14(2):105-21. doi: 10.1016/0309-1740(85)90086-5. |
|
[Robins] |
Robins, S. P., Brady, J. D., “Collagen Cross-Linking and Metabolism”. doi: 10.1016/B978-0-12-373884-4.00035-5. In: “Principles of Bone Biology”, 3.rd Edition, Volume I, 2008, Pages 319-334; ISBN: 978-0-12-373884-4. |
|
[Rosenberg] |
Rosenberg, S. et al., “Further Studies in the Use of Gelatin in the Treatment of Brittle Nails”, Arch Derm. 1957;76(3):330-335. doi: 10.1001/archderm.1957.01550210054006. |
|
[Scala] |
Scala J. et al., “Effect of daily gelatin ingestion on human scalp hair”, Nutr. Report Inter., 13, 579-592 (1976) |
|
[Shenoy] |
Shenoy, M et al. (May 09, 2022), “Collagen Structure, Synthesis, and Its Applications: A Systematic Review”. Cureus 14(5): e24856. doi:10.7759/cureus.24856. |
|
[Shigemura] |
Shigemura, Y. et al., “Identification of a novel food-derived collagen peptide, hydroxyprolyl-glycine, in human peripheral blood by pre-column derivatisation with phenyl isothiocyanate”, Food Chem. 2011, 129, 1019−1024. doi: 10.1016/j.foodchem.2011.05.066. |
|
[Shigemura2] |
Shigemura, Y. et al., “Effect of prolylhydroxyproline (Pro-Hyp), a food-derived collagen peptide in human blood, on growth of fibroblasts from mouse skin”, J. Agric. Food Chem. 2009, 57, 444−449. doi: 10.1021/jf802785h. |
|
[Shigemura3] |
Shigemura, Y. et al., “Changes in Composition and Content of Food-Derived Peptide in Human Blood after Daily Ingestion of Collagen Hydrolysate for 4 weeks”, J Sci Food Agric. 2018 Mar; 98(5):1944-1950. doi: 10.1002/jsfa.8677. |
|
[Shigemura4] |
Shigemura, Y. et al., “Dose-dependent changes in the levels of free and peptide forms of hydroxyproline in human plasma after collagen hydrolysate ingestion”, Food Chem., 159 (15), September 2014, Pages 328-332. doi: 10.1016/j.foodchem.2014.02.091. |
|
[Shoulders] |
Shoulders, M. D., “Collagen structure and stability”, Annu Rev Biochem. 2009:78:929-58. doi: 10.1146/annurev.biochem.77.032207.120833. |
|
[Silvestrini] |
Silvestrini, B. et al., “Collagen Involvement in Health, Disease, and Medicine”, IntechOpen (2022). doi: 10.5772/intechopen.101978. |
|
[Sripriya] |
Sripriya, R. et al., “A Novel Enzymatic Method for Preparation and Characterization of Collagen Film from Swim Bladder of Fish Rohu (Labeo rohita)”, Food and Nutrition Sciences, 2015, 06(15), Pages 1468-1478, Article ID:61421. doi: 10.4236/fns.2015.615151 |
|
[Sugihara] |
Sugihara, F. et al., “Quantification of hydroxyprolyl-glycine (Hyp-Gly) in human blood after ingestion of collagen hydrolysate”, J Biosci Bioeng. 2012 Feb;113(2):202-3. doi: 10.1016/j.jbiosc.2011.09.024. |
|
[Sutmuller] |
M. Sutmuller e al., “Collagen types Vlll and X, two non-fibrillar, short-chain collagens. Structure homologies, functions and involvement in pathology”, Histol Histopathol (1997) 12: 557-566. |
|
[Szoka] |
Szoka, L. et al.,“Exogenous proline stimulates type I collagen and HIF-1α expression and the process is attenuated by glutamine in human skin fibroblasts”, Mol Cell Biochem. 2017 Nov;435(1-2):197-206. doi: 10.1007/s11010-017-3069-y. |
|
[Taga] |
Taga, Y. et al., “Highly Accurate Quantification of Hydroxyproline-Containing Peptides in Blood Using a Protease Digest of Stable Isotope-Labeled Collagen”, J. Agric. Food Chem. 2014, 62, 12096−12102. doi: dx.doi.org/10.1021/jf5039597. |
|
[Tanaka] |
Tanaka, M. et al., “Effects of collagen peptide ingestion on UV-B-induced skin damage”, Biosci Biotechnol Biochem. 2009 Apr 23;73(4):930-2. doi: 10.1271/bbb.80649. |
|
[TCMWiki] |
https://tcmwiki.com/ |
|
[Wang] |
Wang, C., “Human lysyl hydroxylase isoforms: multifunctionality of human LH3 and the amino acids important for its collagen glycosyltransferase activities”, Academic Dissertation, Faculty of Science, University of Oulu, ISBN:9514267990; retrieved at https://urn.fi/URN:ISBN:9514267990] |
|
[WangX] |
Wang X. Et al., “Off-flavour compounds in collagen peptides from fish: Formation, detection and removal”, International Journal of Food Science and Technology 2023, 58, 1543–1563. doi: 10.1111/ijfs.15962. |
|
[Watford] |
Watford, M., “Glutamine metabolism and function in relation to proline synthesis and the safety of glutamine and proline supplementation”, J Nutr. 2008 Oct;138(10):2003S-2007S. doi: 10.1093/jn/138.10.2003S. |
|
[Whitmore] |
Whitmore, R. et al., “Digestibility and safety of limed hide collagen in rat feeding experiments”. J. Food Sci. 1975, 40, 101. doi: 10.1111/j.1365-2621.1975.tb03746.x. |
|
[Wikipedia] |
https://it.wikipedia.org and https://en.wikipedia.org |
|
[Wu] |
Wu, Lingyu et al., “Dietary gelatin enhances non-heme iron absorption possibly via regulation of systemic iron homeostasis in rats”, Journal of Functional Foods (2019) 59, 272-280. doi: 10.1016/j.jff.2019.06.005. |
|
[Xu] |
Xu, Q. et al., “Bioavailability of bioactive peptides derived from food proteins across the intestinal epithelial membrane: A review”, Trends in Food Sci Technol, 86, April 2019, Pages 399-411. doi: 10.1016/j.tifs.2019.02.050 |
|
[Yamada] |
Yamada, S. et al., “Functional roles of fish collagen peptides on bone regeneration”, Dent Mater J. 2021 Dec 1;40(6):1295-1302. doi: 10.4012/dmj.2020-446. |
|
[Zhao] |
Zhao, Zhongjie et al., “阿胶、新阿胶、黄明胶、马皮胶、杂皮胶的质量对比研究 —Ⅰ.氨基酸分析 [Comparative Study on the Quality of E Jiao, New E Jiao, Huang Ming Jiao, Horse Hide Gelatine, and Miscellaneous Hide Gelatine—I. Amino Acid Analysis]”, 天然产物研究与开发 (Natural Product Research and Development) 1991, 3: 49–59. doi:10.16333/j.1001-6880.1991.03.009. |
|
[Zhu] |
Zhu S, Huang M, Feng G, et al., “Gelatin versus its two major degradation products, prolylhydroxyproline and glycine, as supportive therapy in experimental colitis in mice”. Food Sci Nutr. 2018;6:1023–1031. https://doi.org/10.1002/fsn3.639. |
Notes
1 Tryptophan is completely absent; cystine and cysteine are present in very small quantities, approximately 1 amino acid residue in 1000.
2 Telopeptide moieties are responsible for some of collagen’s immunogenic properties. Treatment with pepsin in an acidic environment allows the telopeptide sequences to be cleaved from tropocollagen, thus obtaining atelocollagen. This process preserves the triple helical structure. Atelocollagen is characterized by greater water solubility than tropocollagen and the lack of immunogenic properties. Atelocollagen, often simply called “collagen,” is widely used to prepare industrial materials and for biological research, particularly for human use.
3 Hyp is found as either trans-4-Hydroxy-L-proline (4Hyp), when proline is hydroxylated by prolyl 4-hydroxylase (P4H), or, at a much lesser extent, as trans-3-hydroxy-l-proline (3Hyp), when the proline is hydroxylated by prolyl 3-hydroxylase (P3H). Their ratio is approximately 100:1. 3Hyp is absent from mammalian (such as human and bovine) type III collagens, present as only 1–2 residues of 3Hyp per chain in types I and II collagen, 3–6 residues per chain in types V and XI collagens, and about 10% of the total hydroxyprolines in type IV collagens of basement membranes. [Hu, Hulmes]
4 In scurvy, the lack of hydroxylation of prolines and lysines causes a looser triple helix.
5 Recent work has shown that both glycosyl transferase activities resides in the multifunctional enzyme LH3 (resident in the endoplasmic reticulum) which, in addition to being a lysyl hydroxylase, is thus also a galactosyltransferase (GT) and a glucosyltransferase (GGT). LH3 is therefore able to catalyze the three consecutive reactions required for the formation of glucosylgalactosyl-hydroxylysine. [Hulmes, Wang]
6 α chains are indicated as αi(T), where T is the type of collagen (i.e., I for type I collagen), and i is 1, 2, …, n and denotes the kind of α chain within the T collagen type. For instance, α1(I) is the first kind of α chain in the collagen type 1, α2(I) being the second kind. Type I collagen contains two identical α1(I) chains and a single, different, α2(I), whereas type III collagen only contains α1(III) chains, being an homotrimeric collagen.
7 Apparent digestibility is a measure of the amount of protein intake (%) available for absorption and is usually calculated by estimation of fecal nitrogen and corresponding dietary intake:
Apparent digestibility = (dietary nitrogen − fecal nitrogen) × 100 / dietary nitrogen.
8 A by-product of leather processing. Liming (that is, treatment with lime, calcium hydroxide) partially hydrolyzes covalent bonds of fibrous collagen.
9 From pigskins that were collected from 6-month-old pigs that had been scalded in water at 60°C for 10 min according to slaughterhouse procedures. [Reuterswärd]
10 An immortalized cell line of human colorectal adenocarcinoma cells.
11 Other than in collagen, hydroxyproline is very found in few proteins. Plants, which lack collagen, contain much less hydroxyproline than animals. [Hu]
12 In the body, the majority of Pro is synthesized from L-glutamate, which in turn is produced from Gln. Within the enterocyte, Gln is degraded to glutamate, which is then transformaed to glutamate-γ-semialdehyde via pyrroline 5-carboxylate (P5C) synthase. Glutamate-γ-semialdehyde spontaneously yields P5C that is then reduced to proline by P5C reductase. Proline is also formed in the body from arginine and ornithine which, through the action of ornithine aminotransferase, yield glutamate-γ-semialdehyde and then Pro via P5C reductase. The expression of P5C synthase is restricted to the intestine, however, and therefore this organ is the only site in the body where glutamine (glutamate) is a precursor of Pro synthesis. In rats, ∼7% of glutamine carbon metabolized within the rat small intestine was used for proline synthesis. [Watford]
13 The gene responsible for the synthesis of the pro-α1 chain in type I collagen.
14While Pro-Hyp is the major peptide found in blood after consuming any collagen type, Hyp-Gly has been found after ingesting hydrolyzed collagen made from at least fish scale and pigskin (see for example [Shigemura, Shigemura3, Sugihara]).
Cover photo credit: Ernesto Rodriguez from Pixabay




